|
Relapses can have long-term effects on the central nervous system, including gait and vision challenges. In children with relapsing disease, studies suggest use of intravenous immunoglobulin (IVIG) and/or disease modifying therapies (DMTs). However, patients who experience more severe attacks and relapsing MOGAD are at risk for more severe deficits. Complete recovery from the onset attack is common in children, and in general the outcome is favorable. Most patients respond well to intravenous steroids, with some also requiring oral prednisone for one to four weeks. In this paper, we propose for the first time indications for MOG-IgG testing based on expert consensus. Disease modifying therapies (DMTs) including therapies for children with relapsing forms of MOGAD.MOG-IgG serum concentrations depend on disease activity (with higher median concentrations during acute attacks than during remission) and treatment status (with lower concentrations while on immunosuppression) and may transiently vanish after plasma exchange if MOG-IgG is negative but MOG-EM still suspected, re-testing during acute attacks, during treatment-free intervals, or 1–3 months after plasma exchange (or IVIG b) is recommended – N.B.At the Neuroscience Center, our goal is to stop symptoms and restore a child's normal growth and development with as few treatment-related side effects as possible. Immunoglobulin class detected, assay type, antigenic substrate and biomaterial used, titer/concentration/units, assay-specific cut-offs and performing laboratory should all be documented (e.g., “Serum MOG-IgG 1:1280 ”)Īs with all laboratory tests, positive test results should always be interpreted in the context of the patient’s overall presentation if “red flags” as defined in Table 4 are present, re-testing of the positive serum sample (or, if not anymore available, at least testing of a follow-up serum sample) is recommended to reduce the potential risk of reproducing false-positive results due to issues inherent to the very method employed, use of a second (and, in the case of discrepant results, third) methodologically different cell-based assay is advisable if in doubt, seek expert advice from a specialized center Testing for MOG-IgM and/or MOG-IgA: Currently not recommended additional MOG-IgM and MOG-IgA antibodies have been described in some MOG-IgG-positive patients the clinical relevance of isolated MOG-IgM or -IgA results is unknown testing for antibodies of the IgM class requires removal of total IgG from the sample to avoid both false-negative (due to high-affinity IgG displacing IgM) and false-positive (due to IgM anti-IgG Fc rheumatoid factors) results Serum: Recommended (specimen of choice) shipment at 4 ☌ or on dry ice advisable if samples do no arrive within 1–2 daysĬerebrospinal fluid: Not usually required, since MOG-IgG is produced mostly extrathecally, resulting in lower CSF than serum titers potentially helpful in rare, selected cases (e.g., strong background due to co-existing high-titer non-MOG serum antibodies) shipment at 4 ☌ or on dry ice advisable Peptide-based ELISA, Western blot: Insufficiently specific, obsolete 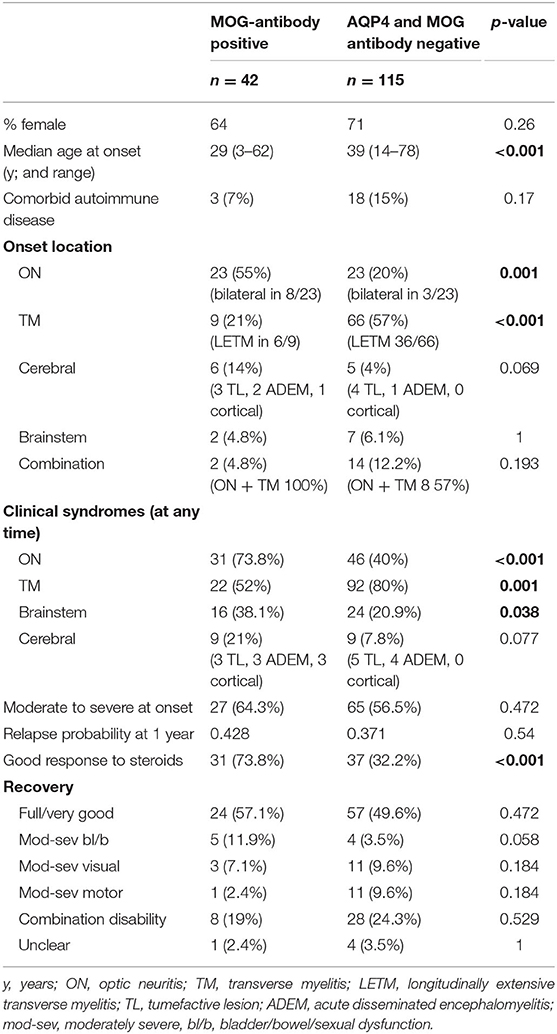
Immunohistochemistry: Currently not recommended (less sensitive than cell-based assays, limited data available on specificity, sensitivity depends on tissue donor species ) if used, Fc-specific secondary antibodies adsorbed against tissue donor IgG required in order to avoid cross-reactivity with IgM and IgA or with tissue-bound donor IgG Test Overview : This panel includes antibodies against anti-myelin oligodendrocyte glycoprotein (MOG) and Aquaporin4 (AQP4) and is used to help establish. MOG-AD is an inflammatory demyelinating condition of the CNS characterised by a monophasic or relapsing course of neurological dysfunction, which does not meet the typical criteria for MS or other known neuroinflammatory conditions and occurs in the presence of serum MOG antibodies detected using specific cell-based assays. Cell-based assays (IFT/FACS): Recommended (current gold standard) must employ full-length human MOG as target antigen use of Fc-specific (or IgG1-specific ) secondary antibodies highly recommended to avoid cross-reactivity with (specifically or non-specifically co-binding) IgM and IgA antibodies Clinical features of MOG antibody disease.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
